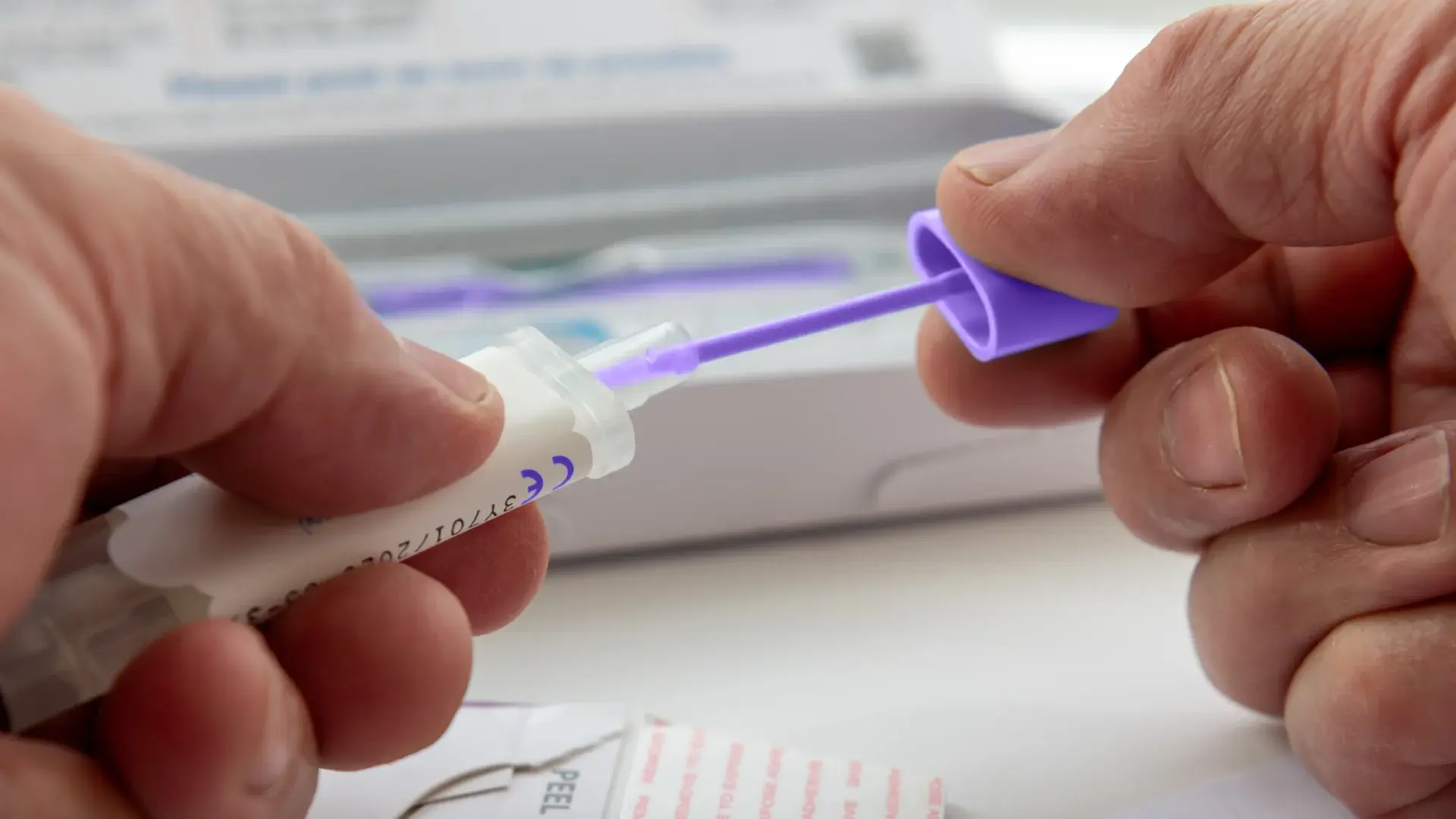
Fraud, waste, and abuse (FWA) schemes involving diagnostic testing continue to evolve, often leveraging complex coverage rules and aggressive outreach practices. Colon cancer screening kits, while clinically valuable when used appropriately, have increasingly appeared in investigations tied to excessive billing, insufficient documentation, and questionable ordering practices.
In this edition of FWA Insights, we examine how Cotiviti’s investigators identified outlier billing patterns from one provider, what their investigation discovered, and how health plans can recognize signs of FWA in home screening tests.
Analyzing abnormal billing for screening tests
A Cotiviti investigator conducted data analysis focused on abnormal billing patterns related to genetic and oncology-related screening tests. The analysis identified a provider with excessive billing tied exclusively to a single genetic test: CPT code 81528, a DNA-based home stool test known as Cologuard®.
Notably, the provider was the only entity submitting claims for CPT code 81528 to the health plan during the review period. This lack of peer comparison raised immediate concerns and warranted further scrutiny. Additional red flags emerged through member complaints. Patients reported being sent colon cancer screening kits they did not request, followed by repeated outreach pressuring them to complete and return the kits. These grievances suggested the testing was not initiated through traditional provider-patient encounters and may not have been supported by documented medical necessity.
Suspicious billing pattern identified
Cotiviti’s special investigations unit (SIU) began by reviewing 24 months of claims data for the identified provider. This review confirmed a pattern of excessive billing involving CPT code 81528 with no evidence of diversified testing or related evaluation and management services. In fact, the claims history showed that the provider solely billed this code during the analysis period.
The investigators identified the provider as an outlier in terms of volume and billing concentration. This comparative analysis reinforced concerns that the provider’s billing behavior did not align with expected clinical practice patterns for colon cancer screening.
To further understand the context, the investigator conducted research related to colon cancer screening kits. Clinical guidelines generally recommend colorectal cancer screening for patients between the ages of 45 and 75 who are at average risk. However, research also highlights several challenges associated with these screenings, including high costs, false positive results, and the potential need for follow-up colonoscopies. Under the Affordable Care Act, routine screenings are typically covered, but if a screening test returns a positive result, the screening may be classified as diagnostic and may not be fully covered by insurance. This shift can result in significant out-of-pocket costs for patients.
The investigation also revealed external enforcement history related to the provider. Publicly available information showed the provider agreed to pay over $10 million dollars to resolve allegations of violating the federal Anti-Kickback Statute. The allegations involved offering prepaid Visa gift cards to Medicare and Medicare Advantage beneficiaries as incentives to return colon cancer screening kits, enabling billing to government health programs. This background heightened concerns regarding the provider’s outreach and billing practices.
Medical record review findings
Cotiviti’s medical review team conducted an initial review of 150 sampled claim lines and identified an overall error rate of 100% due to insufficient or missing documentation. The clinical review revealed the following deficiencies:
-
No supporting documentation (such as office or progress notes) to justify the ordering of CPT code 81528
-
Absence of a valid, signed provider order for the billed test
-
No CLIA certificate included in the medical record
As a result of these documentation and compliance failures, the actual overpayment identified totaled more than $77,000. These findings reinforced the importance of combining claims analytics, member complaints, and medical record review when evaluating potential FWA related to diagnostic testing.
Key takeaways for health plan SIUs
This investigation highlights broader risks tied to the use of mailed colon cancer screening kits. While these tests can improve screening access when appropriately ordered, they also present opportunities for misuse. Aggressive or misleading outreach may blur the line between legitimate preventive care and schemes designed to drive billing volume rather than improved clinical outcomes.
To help health plans succeed in catching improper billing related to colon cancer screening kits, we recommend taking a proactive, analytics‑driven approach that includes the following actions:
-
Implement analytics to detect patterns of repeated tests billed without required clinical orders or supporting documentation
-
Flag providers or vendors with unusually high volumes of “cold” test orders or kits shipped without documented medical necessity
-
Monitor for deceptive marketing practices, including sales tactics that are presented as medical outreach
-
Review member complaints that indicate repeated or persistent outreach for tests that were not ordered by the member’s treating provider
Webinar: Waste and abuse in behavioral health
Join Cotiviti’s FWA experts for a discussion on the behavioral health trends having the greatest impact on Medicaid—and what they signal for other lines of business. Attend our webinar on Tuesday, May 19 as we cover:
-
The latest state‑level guidance designed to stop Medicaid fraud and abuse
-
How emerging behavioral health trends are being exploited by bad actors
-
Key takeaways from the recent federal release of behavioral health provider data
-
Tools and approaches to identify and stop FWA without creating provider abrasion